Ashish Joshi1, Kamalpreet Kaur2, Vishakh Saraf2, Arul Prakash2
1CUNY Graduate School of Public Health and Health Policy, New York, USA
2 Foundation of Healthcare Technologies Society, New Delhi, India
Importance of COVID-19 testing data
Tests allow us to identify infected individuals, guiding the medical treatment that they receive. It enables the isolation of those infected and the tracing and quarantining of their contacts. It can help allocate medical resources and staff more efficiently. Informs our understanding of the pandemic and the risks it poses in different populations. Without data on COVID-19, we cannot possibly understand how the pandemic is progressing, cannot respond appropriately to the threat neither as individuals nor as a society. Nor can we learn where countermeasures against the pandemic are working. The number of confirmed cases is what informs us about the development of the pandemic.
Sources of testing data
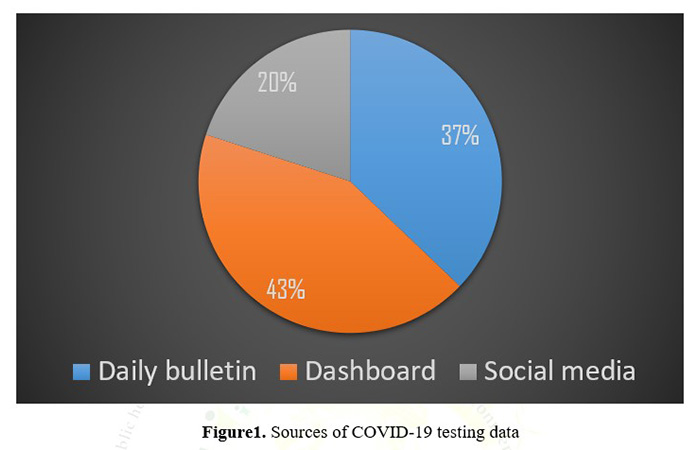
The Indian Council of Medical Research provides a daily bulletin of tests done from all over the country, channeling both private lab data and the conventional government testing centers in both hospitals and BSL accredited labs. The data uploaded every day on the ICMR Website in pdf format, which can be downloaded1. The states and UTs of India have various mechanisms in place for updating their data. However, the timing of releasing the testing data is disparate. Sources of testing data include social media, daily bulletin and Government dashboard. Testing data released through Government dashboard (43%), daily bulletin (37%) and social media (20%) (Figure1)
India has 57, 74,133 sample tested (As of 15-June 2020, 9.00am)1. There are 901 total operational testing laboratories in India. Out of which, 653 are governmental and 248 are private laboratories.
India has definitely strengthened the laboratory testing capacity. Starting with only one COVID-19 lab test at the Indian Council of Medical Research’s National Institute of Virology, Pune in January. Now there are 901 labs across the country and on May 18 2020, India had conducted 100,000 test a day, which was a remarkable achievement2.But with1.36 billion population there is a long way to go.
WHO urges all countries to test and test as testing is important to not only keep people safe but also for the spatial spread of infection. Moreover, quick and timely testing would facilitate the identification and isolation of the infected, which can then be treated effectively3. Testing would also help to analyze the extent and prevalence of the infection, which can guide policy makers to take appropriate steps to contain the infection
Challenges
- India densely populated with 464 people/km2 whereas, countries such as Italy, Spain, Iran and USA has 206, 91, 52 and 36 people/km2 respectively3.With this large population, relatively high and ubiquitous testing rate is required. India has 0.8 doctors per 1000 population and 0.7 hospital beds per 1000 population4.The shortage of beds and doctors and ventilators would further burden the healthcare system.
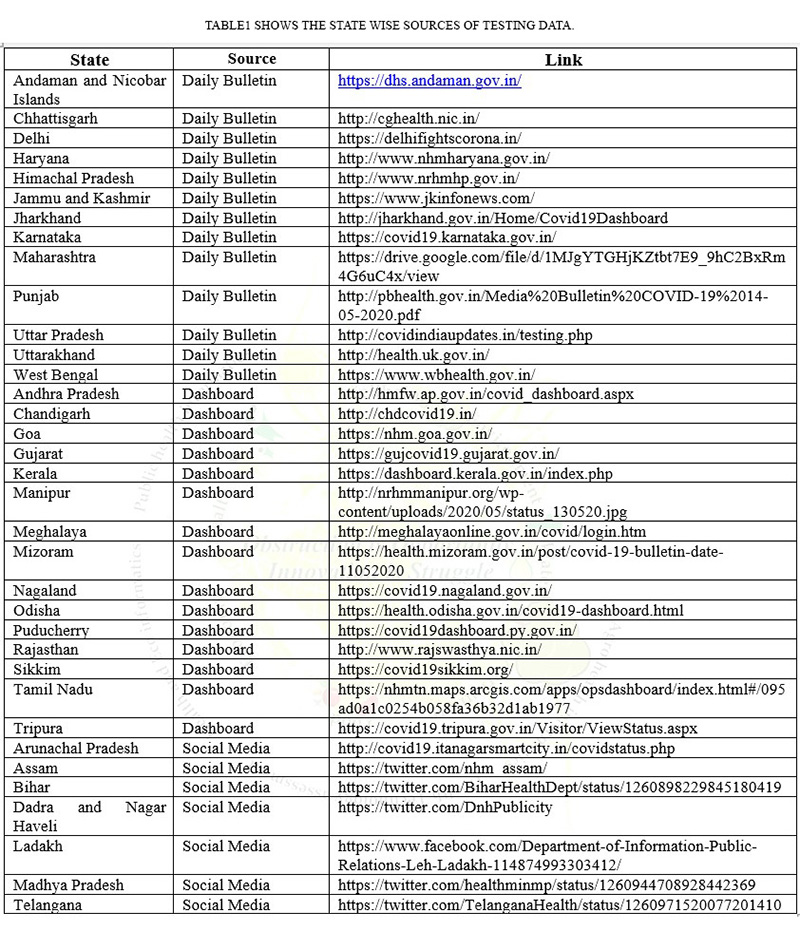
- Currently, India is conducting three types of tests (Real-Time RT PCR, True Nat Test and CBNAAT Test) for COVID-19 but not all of these tests are available in all the states and Union territories5.(Table2 shows the availability of tests in all states and Union territories)
- As shown in above graph (Figure 3), India was conducting fewer test per million population (as on 15 June 2020)6 which is fewer than the countries with highest COVID-19 cases and fatality per million (SMAART Rapid tracker)7. The lower testing means that new cases are underreported and therefore chances of infection could be higher which could become the major public health challenge.
- ICMR recommended using all real-time PCR kits approved by USFDA and European CE and approved by CDSCO. There are RT-PCR kits validated by ICMR-NIV, Pune RT-PCR8. One of the drawback of RT-PCR is that it takes longer time to produce results. Moreover, the test require well trained lab professional as poor-quality specimens, late or very early collection, improper handling of the sample, and inaccurate testing methods can produce COVID-19 negative result9,10.While performing this test false positive and false negative chances should also be considered.
- With Cartridge Based Nucleic Acid Amplification Test (CBNAAT), using Cepheid Xpert Xpress SARS-CoV2 ICMR recommends carried under Biosafety 2 level (BSL-2) conditions and with appropriate biosafety precautions and well-trained microbiologist is required11.
- Another challenge that needs addressed is migrant population who moved out from their current location to their native places during complete lock down. Due to this possibility of exposure to the virus was high. It is imperative that worker tested at source, in transit and at destination12, which in turn requires systematic public health approach.
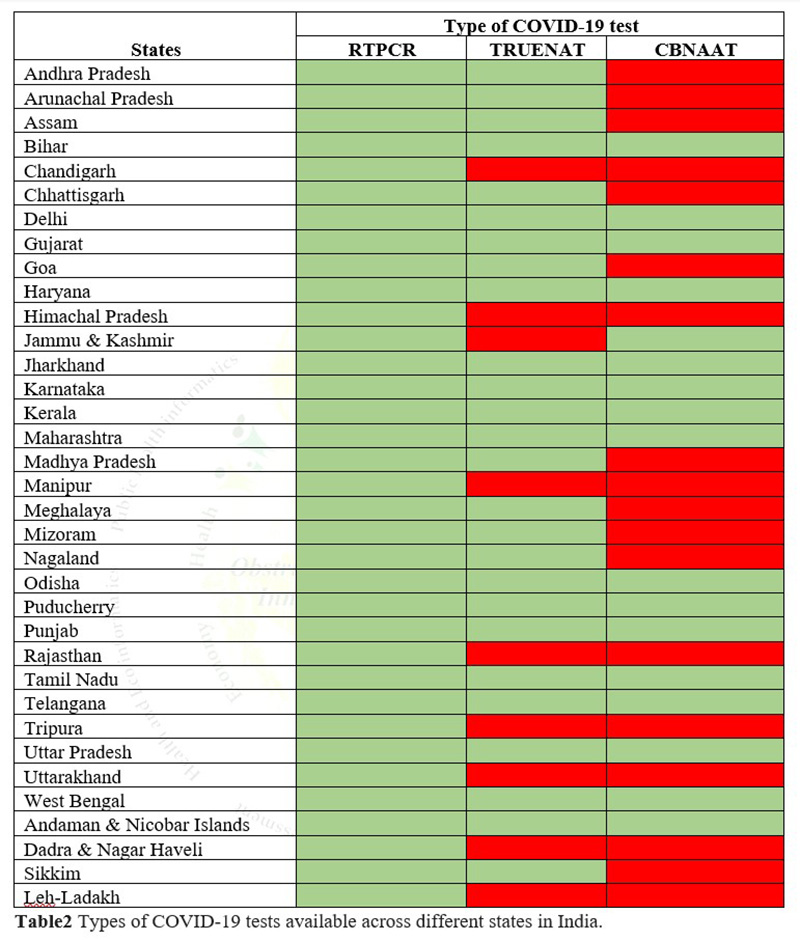

Green color: Test available
Red color: Test not available
Opportunities
- Although, informal settlements are least prepared for the pandemic but for future directions, long term plans, policies, disaster plans should be laid out for the population living in informal settlements by taking the knowledge forward from previous and current pandemics12
- Rapidly diagnostic testing kits demand is on surge globally, so India should use this challenge as an opportunity and produce indigenous cheaper testing kits, which would in turn lower the cost of current tests and will give boost to biomedical industry.
- Even though, ramping up the lab testing increases the demand of reagents swabs and other supplies but it would also create employment for various sectors.
It is very crucial that countries provides the data on testing in a clearly documented way that inform data driven, evidence-based public policy and also establishes trust among citizens to understand the published data and for epidemiologists to incorporate the data into models for building better predictive models.
References
- https://www.icmr.gov.in/
- https://www.icmr.gov.in/pdf/press_realease_files/ICMR_Press_Release_India_testing_story_20052020.pdf
- Kamath S, Kamath R, Salins PCOVID-19 pandemic in India: challenges and silver liningsPostgraduate Medical Journal Published Online First: 16 April 2020. doi: 10.1136/postgradmedj-2020-137780
- Physicians(per1,000people)(n.d.)Available: https://data.worldbank.org/indicator/SH.MED.PHYS.ZS
- https://www.icmr.gov.in/pdf/covid/labs/COVID_Testing_Labs_25052020.pdf
- https://www.worldometers.info/coronavirus/#countries
- http://www.smaartrapidtracker.org
- https://www.icmr.gov.in/pdf/covid/labs/Advisory_for_Reagents_TestingLabs_v1.pdf
- Iyer, Mahalaxmi et al. “COVID-19: an update on diagnostic and therapeutic approaches.” BMB reports vol. 53,4 (2020): 191-205. doi:10.5483/BMBRep.2020.53.4.080
- https://main.icmr.nic.in/sites/default/files/upload_documents/Cepheid_Xpert_Xpress_SARS-CoV2_advisory_v2.pdf
- Kurien, Thomas & Bhat, Ramesh &Kurapati, Sudhakar& Rao, Arni. (2020). Addressing Immediate Challenges in Controlling COVID-19 in India.
- Corburn, Jason et al. “Slum Health: Arresting COVID-19 and Improving Well-Being in Urban Informal Settlements.” Journal of urban health: bulletin of the New York Academy of Medicine, 1–10. 24 Apr. 2020, doi:10.1007/s11524-020-00438-6












Leave A Comment